Translate this page into:
Current Endovascular Treatment Options in Acute Pulmonary Embolism

-
Received: ,
Accepted: ,
How to cite this article: Moore K, Kunin J, Alnijoumi M, Nagpal P, Bhat AP. Current endovascular treatment options in acute pulmonary embolism. J Clin Imaging Sci 2021;11:5.
Abstract
Acute pulmonary embolism (PE) is a significant cause of mortality and morbidity across the globe. Over the last few decades, there have been major therapeutic advances in acute PE management, including catheter-based therapy. However, the effectiveness of catheter-based therapy in acute PE is not supported by Level I evidence, making the use of this promising treatment rather controversial and ambiguous. In this paper, we discuss the risk stratification of acute PE and review the medical and endovascular treatment options. We also summarize and review the data supporting the use of endovascular treatment options in acute PE and describe the potential role of the PE response team.
Keywords
Thrombectomy
Massive pulmonary embolism
Pulmonary embolism response team
FlowTriever
Chronic thromboembolic pulmonary hypertension
INTRODUCTION
Acute pulmonary embolism (PE) is a significant cause of morbidity and mortality due to both abrupt hemodynamic and respiratory changes as well as the chronic sequalae of PE.[1,2] Patients who develop dyspnea with functional limitation without identifiable pulmonary vascular disease that persists for longer than 3 months after the treatment of acute PE, chronic thromboembolic pulmonary hypertension (CTEPH), and chronic thromboembolic disease (CTED), and are encompassed within the more broad “post-PE syndrome” terminology.[3] CTEPH occurs in 0.5%–6.1% of patients after acute PE.[4-7] Some of the factors that predispose to the development of CTEPH include insufficient and inappropriate anticoagulation, recurrent thromboembolic events, elevated pulmonary artery pressure at presentation, hypercoagulability due to various conditions, and other medical conditions. Patients with CTED report symptoms such as dyspnea and exercise limitation similar to those with CTEPH; however, the primary difference is that patients with CTED do not have pulmonary hypertension on hemodynamic studies.[3,8,9] The landscape for the management of sub-massive and massive PE is rapidly evolving. Although anticoagulation (AC) is the standard treatment in the management of PE, some patients may require more aggressive management with systemic thrombolysis (ST) or endovascular treatments. However, catheter directed therapy in acute PE is not supported by Level I evidence, making the use of this promising treatment rather controversial and ambiguous. In this paper, we aim to discuss the risk stratification, pathophysiology, and presentation of acute PE and review the medical and endovascular treatment options for this acute situation. We hope to provide data-based clarification for the role of endovascular therapy in the management of acute PE and provide information to help select the patients who would derive the greatest benefit from endovascular therapy.
DISCUSSION
Risk stratification
PE stratification is a crucial step in clinical decision-making. The American Heart Association (AHA)[10] and the European Society of Cardiology (ESC)[11] have published the following stratification schemes;
Massive (AHA) or high risk (ESC)
These patients are hemodynamically unstable (systolic blood pressure [SBP] of <90 mmHg for over 15 min or need vasopressors). This presentation constitutes ~5% of cases, but has the highest 30 day mortality rate exceeding 50%.[10,12]
Sub-massive (AHA) or intermediate risk (ESC)
These patients present with right ventricular (RV) strain as evidenced, by echocardiography/ computed tomography or RV injury as indicated by elevated biomarkers such as troponins or brain natriuretic hormone. The ESC further sub stratifies intermediate risk PE into low and high risk based on a simplified PE severity index (sPESI) score [Table 1].[10,11,13] Patients with a sPESI of one or more with both RV strain and injury are stratified into the Intermediate-HIGH RISK category. Patients with a sPESI of one or more with either RV strain or injury or neither of these are fall under the Intermediate-LOW RISK group. As a group the sub-massive PE patients constitute about 35‒55% of the PE patients.[1,14-16] The mortality rates in this group of patients treated with AC alone, over a period of 90 days ranges from 2% to 15%.[17-25]
| Parameter | Score (point) |
|---|---|
| Age > 80 years | 1 |
| History of Cancer | 1 |
| History of chronic cardiopulmonary disease | 1 |
| Heart rate of > 110 beats per minute | 1 |
| Systolic blood pressure < 100 mmHg | 1 |
| O2 saturation < 90% | 1 |
Low risk (AHA and ESC)
These are hemodynamically stable patients (peak SBP ≥90 mmHg) and have no RV dysfunction or myocardial injury. Fortunately, majority of the patients fall into the low-risk category and have an excellent prognosis. Most recent guidelines, from various chest and cardiac societies, recommend the addition of ST to standard AC for massive PE.[11,26-28] However, the recommendations for the use of adjunct procedures/treatments in sub-massive PE are less specific. The guidelines or expert consensus do not address the use of advanced endovascular treatment options or the clinical circumstances in which they may be appropriate. To address such ambiguity and to close the gap in practice variability, PE response teams (PERT) have been introduced in some hospitals.[29]
PERT
In 2012, the Massachusetts General Hospital introduced PERT.[30] The function of a PERT is to respond promptly in the situation of an acute PE, assess the patient, come up with a treatment plan, and implement it. The PERT streamlines multispecialty care and communication, which is critical in management of acute PE. PERT can also ensure outpatient follow-up if needed and serve as a platform for large scale PE registries/trials.
The composition of a PERT can be variable between institutes, but often include physicians (both interventional and non-interventional) from cardiology, pulmonary/ critical care, interventional radiology, diagnostic radiology, emergency medicine, cardio thoracic surgery, hematology, vascular medicine/surgery, and pharmacy.
Risk stratification-based treatment of acute PE
Massive PE
Patients with acute massive PE need to be treated in a critical care setting and often require prompt aggressive measures such as ST, surgical thrombectomy, endovascular catheter based treatment, and/or mechanical circulatory support in addition to AC [Figure 1].[10,11,26,31,32] ST involves infusion of a thrombolytic agent through a peripheral IV. The use of 100 mg of alteplase as a continuous infusion for over 2 h has been approved in the United States by the Food and Drug Administration (FDA) for the treatment of acute massive PE.[33] The additional use of ST with standard AC has been shown to improve survival in patients with massive PE.[17,34,35] Mechanical thrombectomy (MT) can be used in patients with massive PE, when there is inadequate time for ST or it is contraindicated.[10,36]
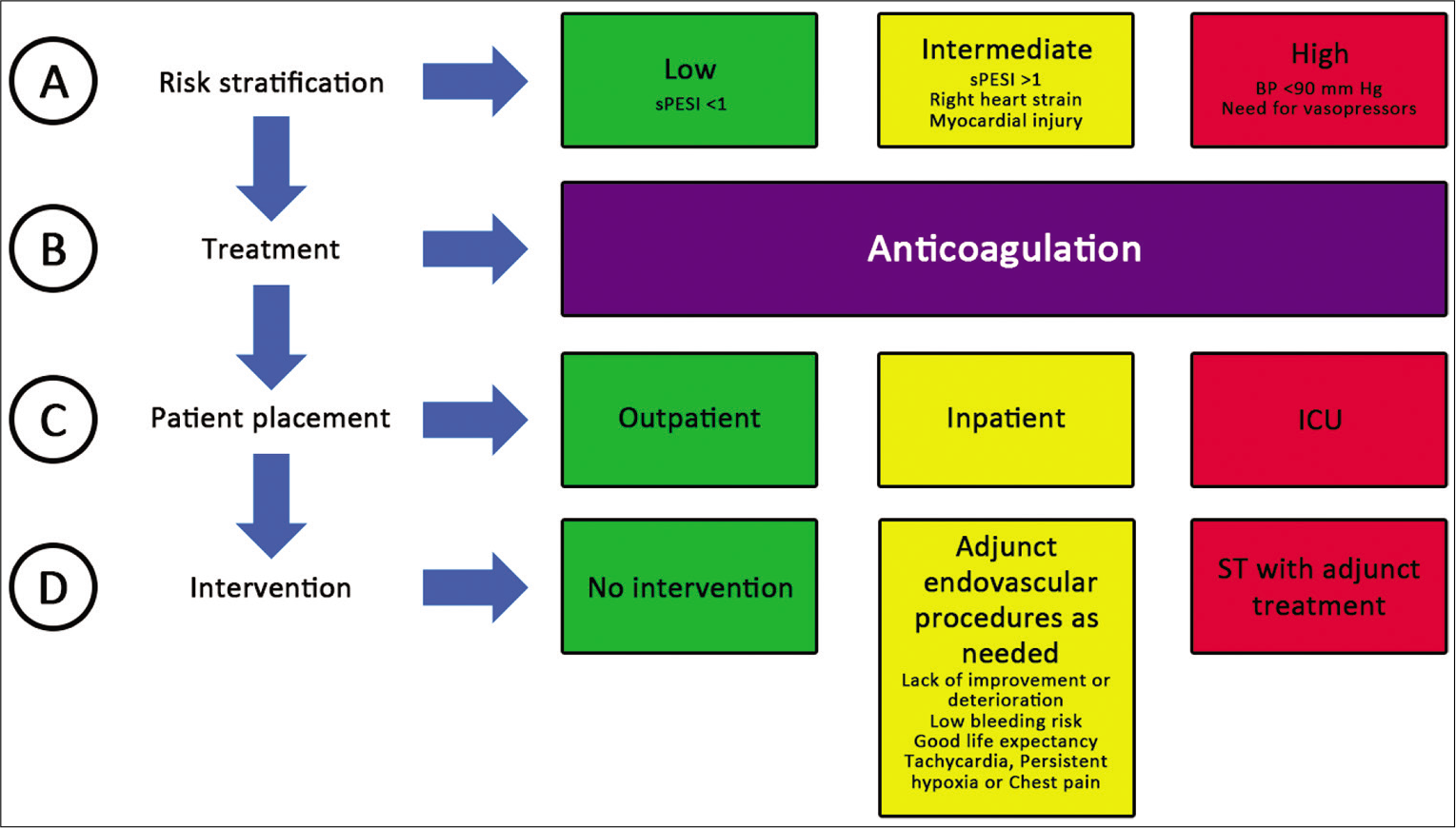
- ABCD of PE management. ST-Systemic thrombolysis, sPESI: Simplified pulmonary embolism severity index, ST: Systemic thrombolysis.
Sub-massive PE
Treatment decisions in sub-massive PE are often the most difficult [Figure 1]. It is unclear if reperfusion therapies in addition to standard AC result in improved overall survival. Predicting which sub-massive PE patients are at a risk of clinical deterioration and will require active intervention is challenging. The benefit of using ST with AC versus AC alone in sub-massive PE has been specifically evaluated by few randomized control trials (RCT).[37,38] These studies did not establish a survival benefit for the addition of ST to AC in the treatment of sub-massive PE. In addition, there have been three meta-analyses evaluating the utility of ST in addition to AC in sub-massive PE. All three studies reported reduced the incidence of clinical deterioration when ST was used as an adjunct to AC when compared to AC alone.[17,39,40] However, only one study reported reduction in overall mortality by addition of ST in the treatment of sub-massive PE.[17] All of the three of the meta-analyses and one of the RCT showed an increased risk of hemorrhagic complications with ST, including fatal intracranial hemorrhage.[17,38-40] In addition, ST along with AC in patients with sub-massive PE may have potential long-term benefits on the RV systolic pressure (RVSP) and exercise capacity.[41]
Based on the available data, it appears that patients receiving ST in addition to AC have a lower risk of acute clinical deterioration when compared to those receiving AC alone, but without proven long-term survival benefit. Potentially, there may be a substantial improvement in RVSP/pulmonary artery pressures long term in patients who receive ST in addition to AC; however, these potential advantages should be weighed against the small but real risk of fatal hemorrhage from using ST.
Low-risk PE
Outpatient AC is the recommended treatment for low-risk PE [Figure 1].
Endovascular treatment for PE
Rationale for intervention
AC alone does not dissolve thrombus, but just prevents propagation of already formed clot. Adverse outcomes in patients with massive and sub-massive PE despite AC have prompted therapeutic escalation through ST, catheter-directed therapies [Figure 2], or surgical/MT [Figure 3]. Patients with evidence of hemodynamic compromise may benefit from the use of hemodynamic support devices such as extracorporeal membranous oxygenation, or isolated percutaneous RV support,[42,43] or advanced endovascular interventions.[44-47]
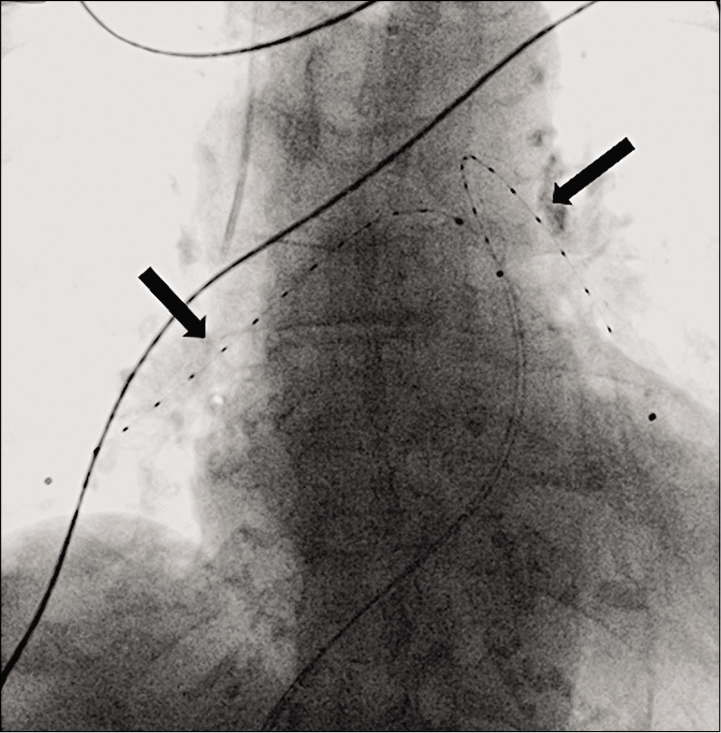
- Chest radiograph of a 60-year-old male with high-risk PE after placement of EKOS catheters in both the pulmonary arteries (black arrows) for the purpose of targeted TPA delivery over 12–24 h.
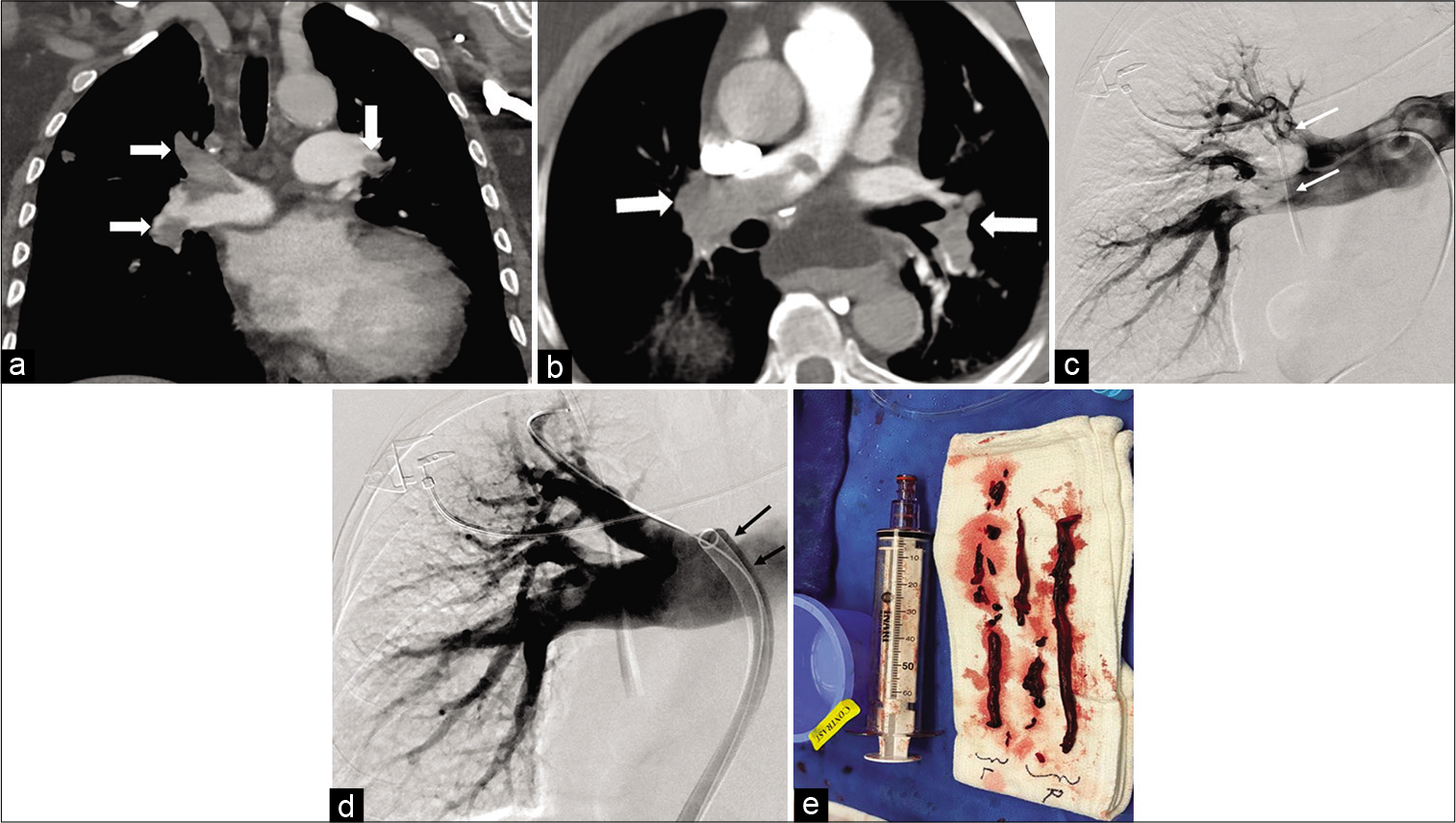
- Coronal (a) and axial CT angiogram (b) of the chest in a 45-year-old male showing bilateral extensive pulmonary embolism (white arrows). Digital subtraction angiogram (DSA) of the right pulmonary artery in the same patient (c) showing multiple filling defects in the upper and lower lobar pulmonary arteries (white arrows). Follow-up DSA (d) of the same patient after mechanical thrombectomy with the FlowTriever device (black arrows) through a 22 F sheath showing near complete clearance of the clot. The aspirated clot is on shown in picture (e) (white arrows).
In patients with sub-massive PE, the primary aim of advanced therapies is to expedite symptom resolution and avert possible migration into the higher risk category resulting from progressive right-sided heart failure.[33] However, there is no Level I evidence supporting improved survival in patients treated with aggressive endovascular therapies versus those treated with standard AC alone.
CTEPH, a rare condition that is found in about 4–6% of patients following acute PE and may require major surgery or balloon angioplasty.[48] Post-PE syndrome (persistent perfusion defects on the lung scan, exercise intolerance and RV dysfunction, however without pulmonary hypertension) occurs in a large proportion of patients recovering from acute PE, despite adequate AC.[49] Preventing these chronic morbidities is another potential but unproven rationale for aggressive endovascular treatments in sub-massive PE.
The decision for active thrombus removal is mainly driven by the severity of the PE but is also influenced by patient preferences, comorbidities, risk factors for major hemorrhage, the site and burden of thrombus, and operator expertise. Table 2 summarizes the mechanism of action, technical details, and regulatory status of some of the commonly used devices.
| Device | Mechanism | Technical consideration | Regulatory status in USA |
|---|---|---|---|
| Ekosonic | USAT+ | 5F catheter | 510 (K) clearance for PA infusion |
| Uifuse | CDL* | 4-5 F catheter | 510(K) clearance for thrombus anywhere in the peripheral circulation |
| Cragg McNamara | CDL | 4-5 F catheter | 510(K) clearance for thrombus anywhere in the peripheral circulation |
| Angio-vac | Veno- Veno bypass | 26 F access for inflow and 16–20 F access for outflow | 510(K) clearance for removal of undesirable intravascular material |
| FlowTriever | Mechanical thrombectomy - suction and maceration of clot | 20/24 F aspiration catheters. Access either through the femoral or jugular vein | 510(K) clearance for PE thrombectomy and clot in transit |
| Indigo | Mechanical thrombectomy -maceration and aspiration | 8/12 F catheters with a suction pump/engine |
510(K) clearance for peripheral thrombectomy |
| Angiojet | Mechanical Rheolytic thrombectomy with optional thrombolysis | 6- 8F catheters attached to a drive unit/pump for thrombectomy and / or infusing TPA |
510(K) clearance for peripheral thrombectomy. Black box warning for use in PA |
Catheter-directed thrombolysis (CDL)
CDL refers to targeted thrombolysis utilizing multi-side hole infusion catheters, to achieve similar or improved effectiveness as ST, however with a lower dose, which, in turn, reduces the risk of major hemorrhage. Dose of TPA used in CDL is approximately 25–50% lower than that given systemically;[50,51] however, the optimal dosing strategy is still being investigated.[52] The Uni-Fuse (AngioDynamics Inc, Latham, NY, USA) and Cragg-McNamara (ev3 Inc, Plymouth, MN, USA) catheters are often used for catheter-based therapy in PE. 4F to 5F catheters with infusion lengths of 5–10 cm are most frequently used. A special dual lumen catheter which uses ultrasound-assisted thrombolysis (USAT) (EKOS Corp, Bothell, WA, USA), [Figure 4] is preferred by some operators for catheter-based treatment of acute PE. One lumen houses a filament/core that emits high-frequency ultrasound, whereas the other lumen infuses thrombolytics through multiple holes along its length. The sound waves enable the dissociation of fibrin strands, and allows exposure of the thrombus ultrastructure to the TPA, thus facilitating better thrombolysis at lower doses of the drug.[53] Infusion of the thrombolytic drug is initiated after placing the catheters in one or both pulmonary arteries, and typically maintained for over 12 h, although more recent data suggest that as little as 2–4 h may have comparable effectiveness.[52] Theoretically, USAT allows for more effective penetration of the thrombolytic drug when compared to standard CDL; however, there are no RCTs to support this claim and in fact some of the available data do not support the additional benefit from use of USA.[54-56]

- EKOS catheter (black arrow heads) with the ultrasound core wire (black arrow). Image provided courtesy of Boston Scientific. ©2020 Boston Scientific Corporation or its affiliates. All rights reserved.
MT
MT includes suction catheters, or other tools to eliminate or reduce the thromboembolic burden in the pulmonary arteries and improve RV afterload without the need for thrombolytics. In massive PE, MT can be used as the sole therapy in patients with contraindications to thrombolytics or as an adjunct to thrombolytic drugs. Only the commonly used techniques are discussed here. High quality selective pulmonary angiograms are typically needed in thrombectomy cases to carefully assess the location of thrombus, potential targets for treatment, and choice of thrombectomy device, which may increase the dose associated with the procedure.[32]
Thrombus maceration
Thrombus can be macerated using a pigtail catheter, guidewire, or appropriately sized balloons. These may be helpful to rapidly establish forward flow and decompress the RV in patients with completely occluded proximal PAs. However, distal embolization may inadvertently result in worsening of hemodynamics.
Rheolytic thrombectomy
The AngioJet device (Boston Scientific, Marlborough, MA, USA) [Figure 5] has been used for PE thrombus removal with variable success. This device uses rapid-speed saline jets directed back from the tip of the catheter, to create a vacuum and facilitate thrombus fragmentation. It comes with a drive unit/pump that creates the high pressure (∼10,000 psi) pulsatile saline flow that exits the catheter tip through multiple retrograde-directed jets and creates a localized low-pressure zone (Venturi effect) for thrombus aspiration and maceration. The catheter can also be used to deliver low-dose thrombolytic agent into the thrombus to aid clot removal. Angio jet driven thrombectomy has been reportedly associated with hypotension, bradycardia, and hypoxia, most likely from byproducts of cell breakdown such as adenosine and bradykinin released into the pulmonary circulation during the procedure. The combination of these complications and the lack of rigorous evidence for this indication has led to a black box warning for use of this device in the pulmonary circulation.[47,57]

- The Angiojet thrombectomy catheter (dotted black arrow) with its dive unit/pump (solid black arrow). Image provided courtesy of Boston Scientific. ©2020 Boston Scientific Corporation or its affiliates. All rights reserved.
Large- and small-bore catheter thrombectomy
The FlowTriever (Inari Medical Inc., Irvine, CA, USA) is the first MT device with FDA indication for the treatment of acute PE. The device has achieved popularity in the US from its simplistic design and more recent publications reporting positive technical success and safety data.[16,58] It consists of a flexible large-bore 20-French (F) aspiration guide catheter, also called T20 [Figure 6a], which tracks over a 0.035-inch stiff guidewire to the level of the clot. The aspiration catheter is introduced through a 22-f sheath placed in the right common femoral vein. Aspiration of the thrombus is performed by applying and subsequently releasing a vacuum that is created manually, by a custom 60 cc syringe [Figure 6b] attached to a side-tube connector.
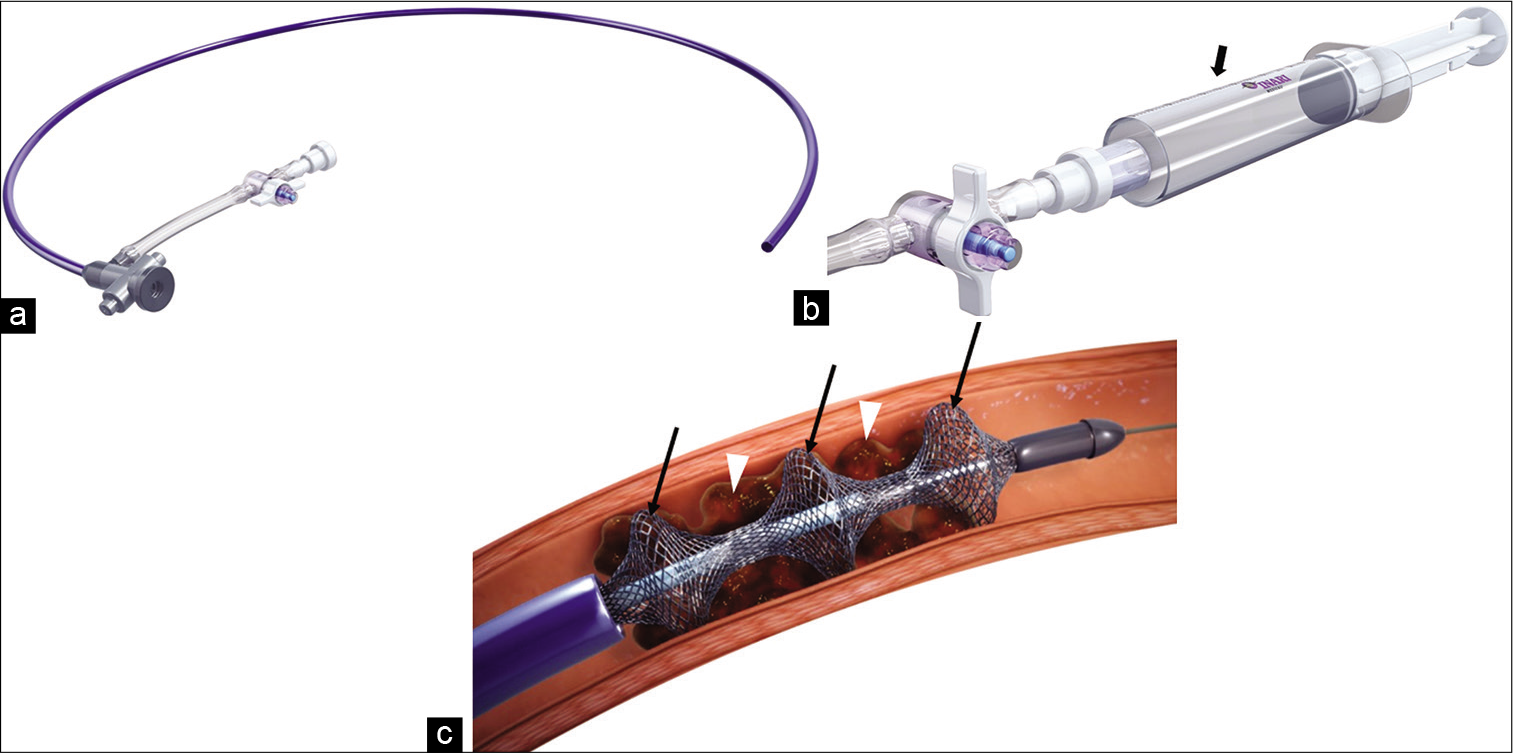
- Picture illustration of the INARI FlowTriever and its components. The 20 F aspiration guide catheter (a), the proprietary syringe (b) (black arrows) and nitinol discs, (c) (black arrows) engaging the clot (white arrow heads). Used with permission from Inari Medical, Irvine, CA.
The FlowTriever system, also comes with self-expanding nitinol discs, in multiple sizes, and can be advanced through the aspiration catheter to promote release of clot attached to the vessel wall [Figure 6c]. More recently a larger version of the aspiration catheter had been released (T24) which needs a larger introducer sheath (24F).
The Indigo Thrombectomy CAT 8 system (Penumbra, Inc, Alameda, CA, USA) consists of an engine that can deliver and maintain vacuum within a flexible 8-French aspiration catheter designed to engage thrombus and extract it [Figure 7a]. The catheter comes with a wire separator to facilitate clot removal [Figure 7b]. The Indigo system lacks the means to return the aspirated blood and this could result in significant blood loss if the catheter is not embedded in the clot. More recently the company launched their CAT 12 system with lightning technology, which is a larger lumen aspiration catheter with technology that helps to vary the amount of suction, based on if the catheter is occlusive on the clot or not and helps optimize clot retrieval while reducing blood loss.
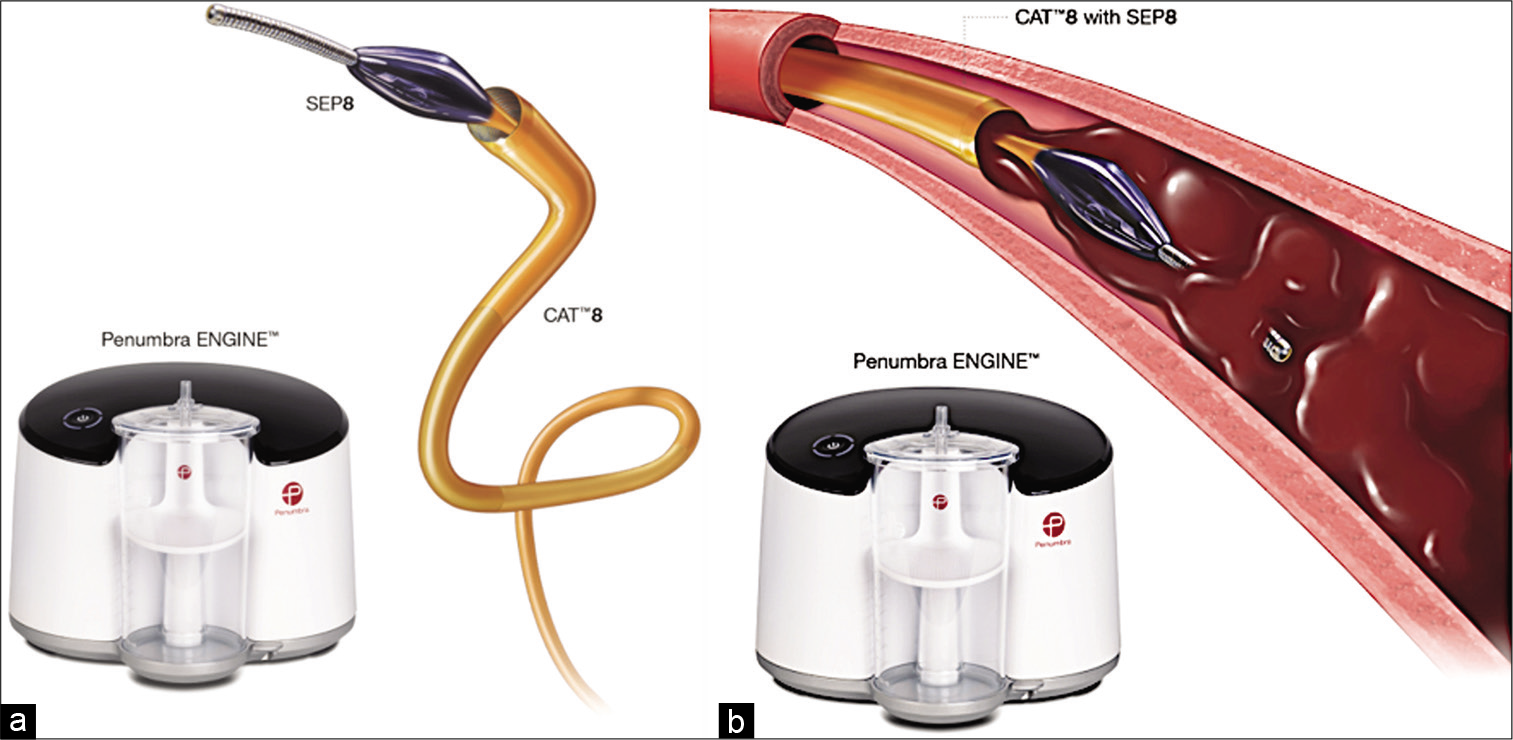
- Illustration (a) of the Indigo CAT8 system with its engine and wire separator (SEP8). (b) CAT8 with deployment of the wire separator (SEP8) in the clot before aspiration. sed with permission from Penumbra, Alameda, CA.
The AngioVac catheter (AngioDynamics Inc., Latham, NY, USA) is a vein-to-vein bypass device intended for retrieval of intravascular material. The inflow cannula is a 22F catheter with a funnel tip, placed in the jugular, or femoral veins. The outflow cannula (16F–20F) returns the blood through a separate access in one of the femoral or jugular veins. An oxygenator can be added to the circuit if needed. Due to its bulk and inflexibility, there have been reports of difficulty in navigating the AngioVac through the right atrium and ventricle into the pulmonary arteries.[59] The device also needs a perfusionist for its operation.
Rotational thrombectomy
Includes devices like the Trerotola (Teleflex Inc., Morrisville, NC, USA) that uses a spinning basket and the Cleaner (Argon Medical Devices, Plano, TX, USA) with a rotating wire, both of which are designed to disrupt thrombus in acute PE. The use of devices has not been extensively reported in patients with PE.
DATA FOR CDL
There have been three prospective studies [Table 3], all utilizing the EKOS catheters and TPA, demonstrating that have demonstrated rapid restoration of RV function.[46,50,51] Although, none of these studies reported fatal intracranial hemorrhage, there was a 10% incidence of major extracranial hemorrhage requiring transfusion in one of the studies.[51] Based on current data it is not clear if long-term clinical outcomes are improved and if CDL it is truly safer than ST. In addition, few meta-analysis and systematic reviews have demonstrated good clinical success for CDL in massive and sub-massive studies ranged from 6‒8% and major bleeding was 3–6%. The mortality rates for massive PE in these studies ranged from 4 to 10%. Given that mortality rates for massive PE in untreated patients is 50%, the low mortality in these studies favors a role for catheter directed treatment in patients with massive PE.
| Study/year | n | Treatment | Control | Follow-up (days) | Submassive PE n(%) | Massive PE n(%) | Results | Complications |
|---|---|---|---|---|---|---|---|---|
| ULTIMA/2013[50] | 59 | tPA-USAT (20 mg) | Heparin | 90 | 59 (100) | 0 (0) | RV/LV ratio reduced from 1.28±0.19 to 0.99±0.17 at 24 h (P<0.001) | 3 minor bleeds |
| SEATTLE II/2015 | 150 | tPA-USAT (24 mg) | No control | 30 | 119 (79) | 31(21) | RV/LV ratio reduced from 1.55 to 1.13 at 48 h (P<0.0001) | 1 major bleed 16 moderate bleeds |
| OPTALYSE PE/ 2018 | 101 | tPA-USAT (8–24 mg) | Compared 4 tPA protocols | 3 | 101 (100) | 0 (0) | RV/LV ratio reduced in all arms | 4 major bleeding 1 recurrent PE 1 death at 30 days and an additional death at 1 year |
CDL: Catheter-directed thrombolysis, GUSTO: Global utilization of streptokinase and tissue plasminogen activator for occluded coronary arteries trial, USAT: Ultrasound assisted thrombolysis, RV: Right ventricle, LV: Left ventricle
DATA FOR MECHANICAL THROMBECTOMY
FlowTriever
In a multicenter prospective single arm trial of 106 patients with acute sub-massive PE (FLARE) [Table 4], the FlowTriever System was found to be safe and effective. The major bleeding complication rate was 1%. One death occurred through the 30-day follow-up, which was not related to the procedure. The study reinforced the significantly reduced need for peri/post-procedure critical care and major complications associated with thrombolytic therapies.[16]
In a separate single-center retrospective study of 46 patients that included both massive and sub-massive central PE with right heart strain, patients treated with the FlowTriever device reported significant reduction of mean pulmonary arterial pressure (MPAP) and oxygen requirements.[58] The major complication rates in this study were less than 5%. No procedure related death was reported [Table 4].
| Study/year | N | Follow-up (days) | Sub massive PE n(%) | Massive PE n(%) | Results | Complications |
|---|---|---|---|---|---|---|
| FLARE/ 2018[16] |
106 | 30 | 104 (100) | 0 (0) | RV/LV ratio 1.53 to 1.15 in 48 h (P<0.0001) | AE-3.8% 1% major hemorrhage 1 death from undiagnosed breast cancer |
| Wible et al./2019 | 46 | 30 | 38 (82) | 8 (18) | Average MPAP before 33.9±8.9 mm Hg; after, 27.0±9.0 mm Hg (P<0.001). 71% experienced reduction in O2 requirements | 1 hemoptysis 1 procedure related blood loss requiring blood transfusion |
| EXTRACT-PE/2020 | 119 | NA | 119 (100) | 0(0) | Reduction in RV/LV ratio: 1.47–1.04. (27.3%). Reduction in systolic PA Pressure: 49–44.5 (9.2%) | MAE 1.7% (2) |
| Yasin et al./2020 |
8 | 90 | 7 (90) | 1 (10) | Reduction in MPAP: 27.8–20.5 (26.3%) | No procedure related adverse events |
PA: Pulmonary arteries, MPAP: Mean pulmonary artery pressure, RV: Right ventricle, LV: Left ventricle, MAE: Major adverse events
Yasin et al. reported a smaller set of patients who were treated with the FlowTriever device with statistically significant decrease in MPAP and O2 requirements without procedure related adverse events [Table 4].[61]
Indigo
A retrospective study of a small number of patients with acute PE using the device (n=6) reported a significant reduction in pulmonary arterial pressure and RV/LV ratio without any major adverse events.[62] A prospective single-arm study (EXTRACT-PE) Evaluating the Safety and Efficacy of the Indigo® Aspiration System in Acute PE just released its results which demonstrated favorable reductions in pulmonary arterial pressure and RV/LV ratios [Table 4].[63]
AngioVac
Data for the use of this device in acute PE comes from case reports and small series that report mixed results.[64-66]
There are no RCT that have been performed to assess the superiority of one device over the others.
CONCLUSION
There has been considerable research into understanding the pathophysiology of acute PE, improving PE risk stratification, and devices to treat acute PE. Although early intervention in sub-massive PE with ST, catheter directed lysis or MT would theoretically prevent escalation to the higher risk category, there are no data to suggest it lowers long-term mortality. Robust high powered prospective RCTs are needed to prove the benefit of these interventions in sub-massive PE. Clinicians and researchers need to look beyond the RV to LV ratio or reduction in MPAP, and study the long-term clinical outcomes following endovascular treatment of acute PE to establish whether clot removal in sub-massive PE yields benefits that offset the risks associated with the treatment. Establishment of PERT may serve as a platform for research into these technologies to ensure optimal use of such devices in the management of acute PE.
Declaration of patient consent
Patient’s consent not required as patients identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflicts of interest
Dr. Nagpal (PN) receives grant support from National Institutes of Health (NIH) and Carver College of Medicine, Iowa City unrelated to this project. The other authors have no conflict.
References
- Acute pulmonary embolism: Clinical outcomes in the international cooperative pulmonary embolism registry (ICOPER) Lancet. 1999;353:1386-9.
- [CrossRef] [Google Scholar]
- Developments in the management and treatment of pulmonary embolism. Eur Respir Rev. 2015;24:484-97.
- [CrossRef] [PubMed] [Google Scholar]
- The Post-Pulmonary Embolism Syndrome: Real or Ruse? 2019 Available from: https://www.atsjournals.org [Last accessed on 2021 Jan 13]
- [CrossRef] [PubMed] [Google Scholar]
- Incidence of chronic thromboembolic pulmonary hypertension after acute pulmonary embolism: A contemporary view of the published literature. Eur Respir J. 2017;49:1601792.
- [CrossRef] [PubMed] [Google Scholar]
- Risk factors and basic mechanisms of chronic thromboembolic pulmonary hypertension: A current understanding. Eur Respiratory Soc. 2013;41:462-8.
- [CrossRef] [PubMed] [Google Scholar]
- Chronic thromboembolic pulmonary hypertension: Evolving therapeutic approaches for operable and inoperable disease. J Am Coll Cardiol. 2018;71:2468-86.
- [CrossRef] [PubMed] [Google Scholar]
- The predictive value of echocardiography for chronic thromboembolic pulmonary hypertension after acute pulmonary embolism in Korea. Korean J Intern Med. 2017;32:85-94.
- [CrossRef] [PubMed] [Google Scholar]
- Chronic thromboembolic disease following pulmonary embolism: Time for a fresh look at old clot. Eur Respir J. 2020;55:1901934.
- [CrossRef] [PubMed] [Google Scholar]
- Preoperative cross-sectional mapping for deep inferior epigastric and profunda artery perforator flaps. Cardiovasc Diagn Ther. 2019;9:S131-42.
- [CrossRef] [PubMed] [Google Scholar]
- Management of massive and submassive pulmonary embolism, iliofemoral deep vein thrombosis, and chronic thromboembolic pulmonary hypertension: A scientific statement from the American heart association. Circulation. 2011;123:1788-830.
- [CrossRef] [PubMed] [Google Scholar]
- ESC guidelines on the diagnosis and management of acute pulmonary embolism: The task force for the diagnosis and management of acute pulmonary embolism of the European society of cardiology (ESC) endorsed by the European respiratory society (ERS) Eur Heart J. 2014;35:3033-80.
- [CrossRef] [PubMed] [Google Scholar]
- Ultrasound-assisted catheter-directed thrombolysis: A novel and promising endovascular therapeutic modality for intermediate-risk pulmonary embolism. Angiology. 2017;68:494-501.
- [CrossRef] [PubMed] [Google Scholar]
- Simplification of the pulmonary embolism severity index for prognostication in patients with acute symptomatic pulmonary embolism. Arch Intern Med. 2010;170:1383-9.
- [CrossRef] [PubMed] [Google Scholar]
- Acute pulmonary embolism: Mortality prediction by the 2014 European society of cardiology risk stratification model. Eur Respir J. 2016;48:780-6.
- [CrossRef] [PubMed] [Google Scholar]
- Management strategies and determinants of outcome in acute major pulmonary embolism: Results of a multicenter registry. J Am Coll Cardiol. 1997;30:1165-71.
- [CrossRef] [Google Scholar]
- A prospective, single-arm, multicenter trial of catheter-directed mechanical thrombectomy for intermediate-risk acute pulmonary embolism: The FLARE study. JACC Cardiovasc Interv. 2019;12:859-69.
- [CrossRef] [PubMed] [Google Scholar]
- Thrombolysis for pulmonary embolism and risk of all-cause mortality, major bleeding, and intracranial hemorrhage: A meta-analysis. JAMA. 2014;311:2414-21.
- [CrossRef] [PubMed] [Google Scholar]
- CT-guided percutaneous neurolytic celiac plexus block technique. Abdom Imaging. 2006;31:710-8.
- [CrossRef] [PubMed] [Google Scholar]
- Predictors of mortality from pulmonary embolism and their influence on clinical management. Thromb Haemost. 2008;100:747-51.
- [CrossRef] [PubMed] [Google Scholar]
- Short-term clinical outcome of patients with acute pulmonary embolism, normal blood pressure, and echocardiographic right ventricular dysfunction. Circulation. 2000;101:2817-22.
- [CrossRef] [PubMed] [Google Scholar]
- The impact of right ventricular dysfunction on the prognosis and therapy of normotensive patients with pulmonary embolism. Chest. 2004;125:1539-45.
- [CrossRef] [PubMed] [Google Scholar]
- Contemporary management and outcomes of patients with massive and submassive pulmonary embolism. Am J Med. 2018;131:1506-14.e0.
- [CrossRef] [PubMed] [Google Scholar]
- Therapy and outcomes in massive pulmonary embolism from the emergency medicine pulmonary embolism in the real world registry. Am J Emerg Med. 2012;30:1774-81.
- [CrossRef] [PubMed] [Google Scholar]
- CT-guided gastrostomy tube placement-a single center case series. Diagn Interv Radiol. 2020;26:464-9.
- [CrossRef] [PubMed] [Google Scholar]
- Simplified rapid protocol for assessing the thoracic aortic dimensions and pathology with noncontrast mr angiography. Int J Angiol. 2019;28:130-6.
- [CrossRef] [PubMed] [Google Scholar]
- Antithrombotic therapy for VTE disease: CHEST guideline and expert panel report. Chest. 2016;149:315-52.
- [CrossRef] [PubMed] [Google Scholar]
- Executive summary: Heart disease and stroke statistics-2014 update: A report from the American heart association. Circulation. 2014;129:399-410.
- [CrossRef] [PubMed] [Google Scholar]
- Role of computed tomography-guided biopsies in the era of electromagnetic navigational bronchoscopy: A retrospective study of factors predicting diagnostic yield in electromagnetic navigational bronchoscopy and computed tomography biopsies. J Clin Imaging Sci. 2020;10:33.
- [CrossRef] [PubMed] [Google Scholar]
- Diversity in the pulmonary embolism response team model: An organizational survey of the national PERT consortium members. Chest. 2016;150:1414-7.
- [CrossRef] [PubMed] [Google Scholar]
- The massachusetts general hospital pulmonary embolism response team (MGH PERT): Creation of a multidisciplinary program to improve care of patients with massive and submassive pulmonary embolism. Hosp Pract 1995. 2014;42:31-7.
- [CrossRef] [PubMed] [Google Scholar]
- Experience with extracorporeal membrane oxygenation in massive and submassive pulmonary embolism in a tertiary care center. Am J Emerg Med. 2013;31:1616-7.
- [CrossRef] [PubMed] [Google Scholar]
- Factors affecting radiation dose in computed tomography angiograms for pulmonary embolism: A retrospective cohort study. J Clin Imaging Sci. 2020;10:74.
- [CrossRef] [PubMed] [Google Scholar]
- Interventional therapies for acute pulmonary embolism: Current status and principles for the development of novel evidence: A scientific statement from the American heart association. Circulation. 2019;140:e774-801.
- [CrossRef] [Google Scholar]
- Thrombolysis compared with heparin for the initial treatment of pulmonary embolism: A meta-analysis of the randomized controlled trials. Circulation. 2004;110:744-9.
- [CrossRef] [PubMed] [Google Scholar]
- The role of an IVC filter retrieval clinic-a single center retrospective analysis. Indian J Radiol Imaging. 2019;29:391-6.
- [CrossRef] [PubMed] [Google Scholar]
- Systemic thrombolysis for pulmonary embolism: Who and how. Tech Vasc Interv Radiol. 2017;20:162-74.
- [CrossRef] [PubMed] [Google Scholar]
- Heparin plus alteplase compared with heparin alone in patients with submassive pulmonary embolism. N Engl J Med. 2002;347:1143-50.
- [CrossRef] [PubMed] [Google Scholar]
- Fibrinolysis for patients with intermediate-risk pulmonary embolism. N Engl J Med. 2014;370:1402-11.
- [CrossRef] [PubMed] [Google Scholar]
- Systemic thrombolytic therapy for acute pulmonary embolism: A systematic review and meta-analysis. Eur Heart J. 2015;36:605-14.
- [CrossRef] [PubMed] [Google Scholar]
- Impact of the efficacy of thrombolytic therapy on the mortality of patients with acute submassive pulmonary embolism: A meta-analysis. J Thromb Haemost. 2014;12:1086-95.
- [CrossRef] [PubMed] [Google Scholar]
- Prospective evaluation of right ventricular function and functional status 6 months after acute submassive pulmonary embolism: Frequency of persistent or subsequent elevation in estimated pulmonary artery pressure. Chest. 2009;136:1202-10.
- [CrossRef] [PubMed] [Google Scholar]
- Extra-corporeal membrane oxygenation and outcomes in massive pulmonary embolism: Two eras at an urban tertiary care hospital. Vasc Med. 2018;23:60-4.
- [CrossRef] [PubMed] [Google Scholar]
- Mechanical circulatory support for high-risk pulmonary embolism. Interv Cardiol Clin. 2018;7:119-28.
- [CrossRef] [PubMed] [Google Scholar]
- A meta-analysis of outcomes of catheter-directed thrombolysis for high-and intermediate-risk pulmonary embolism. J Vasc Surg Venous Lymphat Disord. 2018;6:530-40.
- [CrossRef] [PubMed] [Google Scholar]
- Pulmonary embolism response to fragmentation, embolectomy, and catheter thrombolysis (PERFECT): Initial results from a prospective multicenter registry. Chest. 2015;148:667-73.
- [CrossRef] [PubMed] [Google Scholar]
- Catheter-directed treatment of pulmonary embolism: A systematic review and meta-analysis of modern literature. Clin Appl Thromb Hemost. 2017;23:821-9.
- [CrossRef] [PubMed] [Google Scholar]
- Catheter-directed therapy for the treatment of massive pulmonary embolism: Systematic review and meta-analysis of modern techniques. J Vasc Interv Radiol. 2009;20:1431-40.
- [CrossRef] [PubMed] [Google Scholar]
- Incidence of chronic thromboembolic pulmonary hypertension after pulmonary embolism. N Engl J Med. 2004;350:2257-64.
- [CrossRef] [PubMed] [Google Scholar]
- Late outcomes of pulmonary embolism: The post-PE syndrome. Thromb Res. 2018;164:157-62.
- [CrossRef] [PubMed] [Google Scholar]
- Randomized, controlled trial of ultrasound-assisted catheter-directed thrombolysis for acute intermediate-risk pulmonary embolism. Circulation. 2014;129:479-86.
- [CrossRef] [PubMed] [Google Scholar]
- A prospective, single-arm, multicenter trial of ultrasound-facilitated, catheter-directed, low-dose fibrinolysis for acute massive and submassive pulmonary embolism: The SEATTLE II study. JACC Cardiovasc Interv. 2015;8:1382-92.
- [CrossRef] [PubMed] [Google Scholar]
- A randomized trial of the optimum duration of acoustic pulse thrombolysis procedure in acute intermediate-risk pulmonary embolism: The OPTALYSE PE trial. JACC Cardiovasc Interv. 2018;11:1401-10.
- [CrossRef] [PubMed] [Google Scholar]
- Ultrasound reversibly disaggregates fibrin fibers. Thromb Haemost. 1997;78:1063-8.
- [CrossRef] [PubMed] [Google Scholar]
- Ultrasound-assisted versus conventional catheter-directed thrombolysis for acute pulmonary embolism: A multicenter comparison of patient-centered outcomes. Vasc Med. 2019;24:241-7.
- [CrossRef] [PubMed] [Google Scholar]
- Comparative outcomes of ultrasound-assisted thrombolysis and standard catheter-directed thrombolysis in the treatment of acute pulmonary embolism. Vasc Endovascular Surg. 2016;50:405-10.
- [CrossRef] [PubMed] [Google Scholar]
- Ultrasound-accelerated thrombolysis (USAT) versus standard catheter-directed thrombolysis (CDT) for treatment of pulmonary embolism: A retrospective analysis. Vasc Med. 2019;24:234-40.
- [CrossRef] [PubMed] [Google Scholar]
- AngioJet rheolytic thrombectomy in patients presenting with high-risk pulmonary embolism and cardiogenic shock: A feasibility pilot study. Eurointervention. 2013;8:1419-27.
- [CrossRef] [PubMed] [Google Scholar]
- Safety and efficacy of acute pulmonary embolism treated via large-bore aspiration mechanical thrombectomy using the inari flowtriever device. J Vasc Interv Radiol. 2019;30:1370-5.
- [CrossRef] [PubMed] [Google Scholar]
- Interventional therapy for pulmonary embolism. Methodist Debakey Cardiovasc J. 2016;12:219-24.
- [CrossRef] [PubMed] [Google Scholar]
- Meta-analysis of catheter directed ultrasound-assisted thrombolysis in pulmonary embolism. Am J Cardiol. 2019;124:1470-7.
- [CrossRef] [PubMed] [Google Scholar]
- Technical efficiency, short-term clinical results and safety of a large-bore aspiration catheter in acute pulmonary embolism-a retrospective case study. Lung India. 2020;37:485-90.
- [CrossRef] [PubMed] [Google Scholar]
- Continuous aspiration mechanical thrombectomy for the management of submassive pulmonary embolism: A single-center experience. J Vasc Interv Radiol. 2017;28:1348-52.
- [CrossRef] [PubMed] [Google Scholar]
- Evaluating the Safety and Efficacy of the Indigo® Aspiration System in Acute Pulmonary Embolism-Full Text View. Available from: https://www.clinicaltrials.gov/ct2/show/nct03218566 [Last accessed on 2021 Jan 13]
- [Google Scholar]
- Successful management of acute massive pulmonary embolism using Angiovac suction catheter technique in a hemodynamically unstable patient. Cardiovasc Revasc Med. 2014;15:240-3.
- [CrossRef] [PubMed] [Google Scholar]
- Thrombectomy using suction filtration and veno-venous bypass: Single center experience with a novel device. Catheter Cardiovasc Interv. 2015;86:E81-7.
- [CrossRef] [PubMed] [Google Scholar]
- Early experience with angiovac aspiration in the pulmonary arteries. J Vasc Interv Radiol. 2016;27:730-4.
- [CrossRef] [PubMed] [Google Scholar]







