Translate this page into:
CT Manifestations of Osler-Weber-Rendu Syndrome in Liver: Report of Three Cases
Address for correspondence: Dr. Qu Chuan-Qiang, Department of Neurology, Provincial Hospital Affiliated to Shandong University, Jinan, 250021, China. E-mail: quchuanqiang@sina.com
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Osler-Weber-Rendu syndrome is characterized by widespread telangiectasias. Its clinical manifestations depend on position and scope of the abnormal vessels. The clinical and CT data of 3 patients with Osler-Weber-Rendu syndrome were retrospectively analyzed. CT features reviewed include the change of volume and configuration of liver, presence of tortuous and irregular vessels, opacified vessel mass, arteriovenous shunt, and splenomegaly. CT is helpful for diagnosis, treatment, and follow-up of Osler-Weber-Rendu syndrome.
Keywords
Computed tomography
liver
maximum intensity projection
Osler-Weber-Rendu syndrome
volume rendering technique
INTRODUCTION

Osler-Weber-Rendu syndrome, also known as hereditary hemorrhagic telangiectasia (HHT), is an autosomal dominant disorder. HHT is characterized by widespread telangiectasias, that can involve the skin, mucous membranes, lung, brain, gastrointestinal tract, and/or liver. In addition to epistaxis, gastrointestinal hemorrhage, mucocutaneous telangiectasia, and iron deficiency anemia, there are pulmonary, brain, and hepatic arteriovenous malformations (AVMs) that can cause serious complications.[1] Patients with pulmonary AVMs may have hypoxemia and/or hemorrhage and patients are at risk of hemorrhage and neurological sequelae due to paradoxical embolism. Cerebral AVMs can lead to headache, seizures, ischemia, or devastating hamorrhage. Cerebral AVMs affect more than 10% of HHT patients.[23] The prevalence of hepatic involvement in HHT had been estimated at 8-31% in retrospective studies[4] but in more recent large prospective series the prevalence is higher, ranging between 41% and 78%.[5] Nevertheless, only 8% of the patients with liver involvement in HHT present with symptoms, including high output heart failure, portal hypertension, or biliary disease.[67] Since the symptomatic involvement of the liver in HHT is an extremely infrequent entity, diagnostic imaging may play a fundamental role in the identification of hepatic vascular alterations.
CASE REPORTS
Case 1: A 35-year-old female had suffered from mild pain in the right upper quadrant for 2 years. Hepatic function tests showed alkaline phosphatase (AKP), 156IU/L; γ-glutamiltranspeptidase (γ-GT), 78IU/L; aspartate aminotransferase (AST), alanine aminotransferase (ALT), and hepatic bilirubin were within the normal range. The patient underwent surgery. The enlarged right and middle hepatic arteries were ligated and left hepatic artery was banded.
Case 2: A 35-year-old female suffered from pain in the right upper quadrant for 8 years. Pigmentation was found on the skin of the right upper abdomen. The lower margin of the liver was 3 cm below the costal margins. Liver function tests showed AKP, 183IU/L; γ-GT, 106IU/L; ALT, AST and hepatic bilirubin were also within the normal range.
Case 3: A 61-year-old male had come for a routine physical examination and had no clinical symptom. Cirrhosis and abnormal tortuous vessels were identified in porta hepatis on ultrasound imaging.
All patients were examined in the supine position on 16-slice spiral of CT SIEMENS SOMATOM Sensation 16. The patients fasted for 8 hours and drank about 700-1000 mL warm water or iodic contrast agent (25%, compound Meglumine Diatrizoate Injection) before examination. The scanning parameters were: slice thickness, 5 mm; pitch, 1.5. Iohexol (300 mgI/m1) was administrated intravenously with the dose 15 mL/kg and the injection ratio 25-30 mL/s.Images were obtained in the arterial, portal venous, and delayed phases. Original images were post-processed at a workstation (SIEMENS Wizard VB10B or GE AW4.2). The volume of the liver was measured with three-dimensional CT reconstruction by the method introduced by Hiroshige.[8]
CT findings
Case 1: Before surgery, the hepatic volume was 2048.235 cm3 measured by 3D CT. The surface of the liver was lobular. The volume of the left lobe to that of the right lobe was disproportionate. On arterial phase, some tortuous and irregular arteries were observed in intra- and extra-hepatic regions, especially in porta hepatis. There was heterogeneous enhancement in hepatic parenchyma with multiple opacified vessel masses and tortuous arteries beneath the capsule of the liver [Figure 1a]. On portovenous phase, the portal system enhanced slightly and the portal and splenic veins were tortuous. The contrast agent filled the superior vena cava and right atrium and no venous varix was found. Tortuous arteries in hepatic parenchyma and vessel masses in the late arterial phase were seen [Figure 1b]. A small amount of ascites was identified around the liver. Splenomegaly was also present. The right atrium was enlarged.
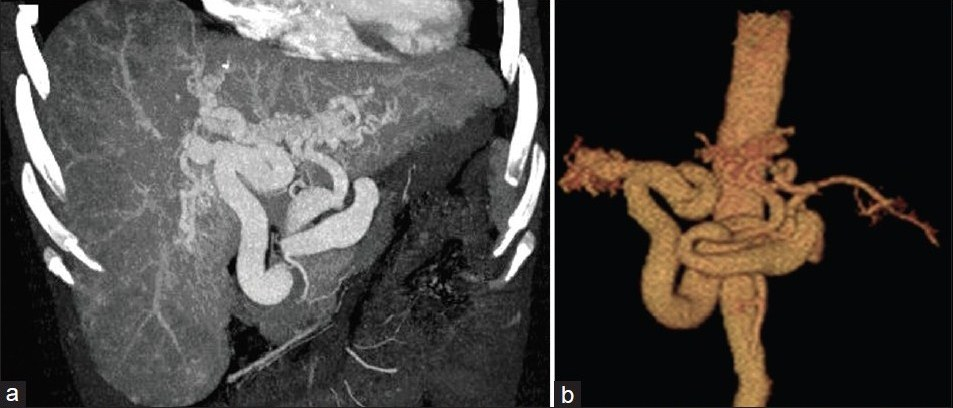
- CT images before surgery in Case 1. Figure 1a is coronal MIP reconstructed image in arterial phase that shows tortuous and irregular arteries in liver and portahepatis. Heterogeneity of the contrast enhancement in hepatic parenchyma with multiple opacified vessel masses is also observed. (b) VRT image shows two tortuous and distended arteries derived from the celiac trunk supplying the left and right lobes of the liver.
One year after surgery, the hepatic volume was 1965.331 cm3 measured by CT. On contrast enhanced CT, MIP, and VRT images [Figures 2a, b] the amount of abnormal arteries decreased in porta hepatis. Telangiectasia in liver also decreased when compared with those before surgery.
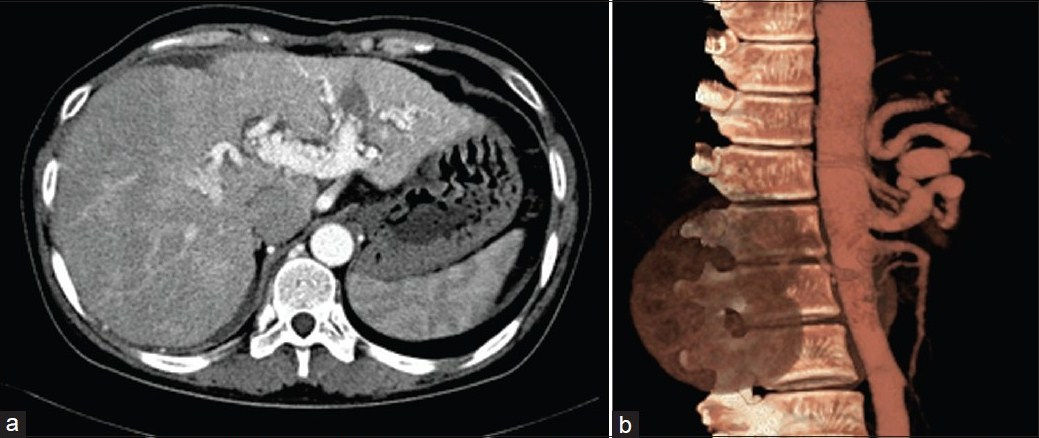
- CT images after surgery in (a) arterial phase and (b) VRT respectively. The arteries in porta hepatis are decreased compared with those before operation.
Case 2: The hepatic volume was 1571.775 cm3. The shape of the liver was irregular. On arterial phase and MIP, VRT images [Figures 3a, b], many tortuous, twisted, and irregular arteries were present in liver parenchyma, porta hepatis, and in front of the abdominal aorta. Varicose veins were identified in the gastric wall, greater omentum, and anterior abdominal wall. On venous phase, portal vein and splenic vein were of normal caliber. The hepatic veins and the right atrium were mildly distended. The intrahepatic biliary ducts were dilated to varying degrees. Mild splenomegaly was present.
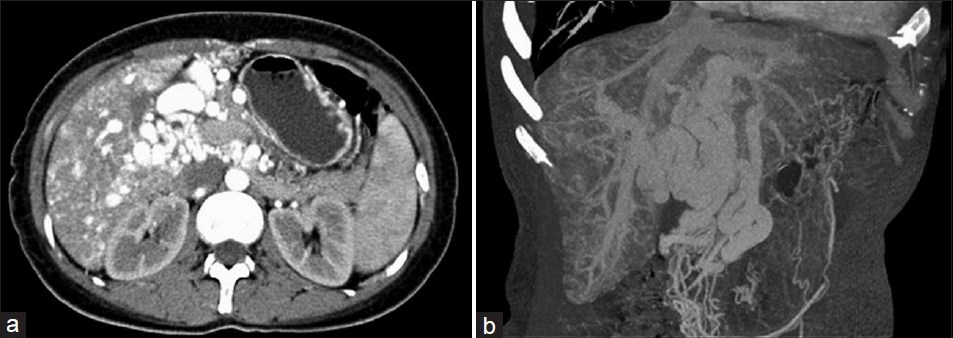
- CT images in arterial phase and VRT respectively in Case 2. Irregular and tortuous arteries in the liver parenchyma, and porta hepatis are present. The intra-hepatic billary ducts are dilated. Splenomegaly with gastric varices is identified.
Case 3: The hepatic volume was 1392.66 cm3. The shape of the liver was abnormal. The caudate lobe was enlarged. On the arterial phase, MIP, and VRT images [Figures 4a, b], celiac trunk was mildly dilated and thicker vessel masses were identified in porta hepatis. On portal venous phase, the intra- and extra-hepatic abnormal vessels were still present, while portal vein and superior vena cava were normal.
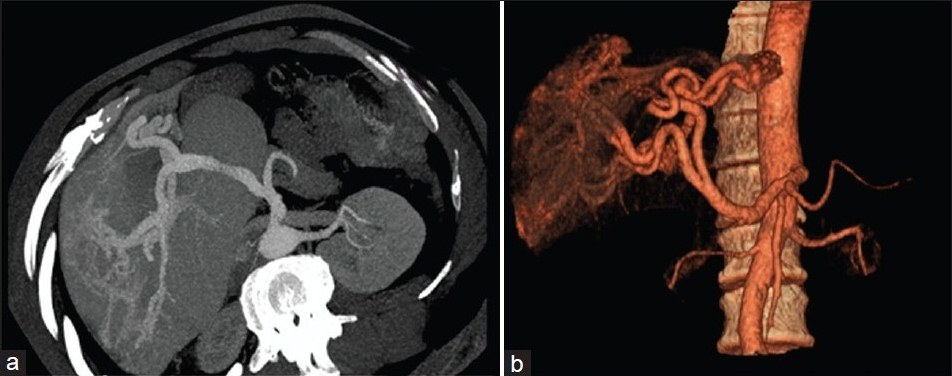
- CT images in arterial phase and VRT respectively in Case 3. Enlarged caudate lobe is present. Tortuous vessels are observed in the porta hepatis.
Summary of Imaging Findings in all 3 patients is summarized in Table 1.
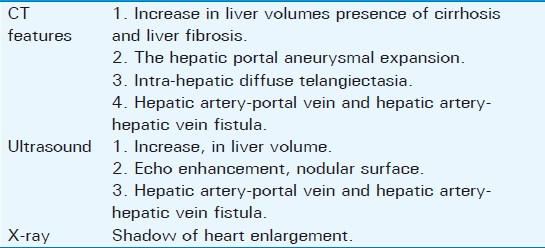
DISCUSSION
Osler-Rendu-Weber syndrome is a relatively rare disease, which was first recognized by Babington in 1865. In 1896, Rendu confirmed its family hereditary, which was proved by Osler and Weber in 1901 and 1907, respectively. This syndrome is an autosomal dominant trait and occurs in any race without gender difference. Its morbidity is about 1/3,500-5,000 in the Western hemisphere,[9] 1/2,351 in France, 1/16,500 in Vermont, 1/350 in Julen, 1/39,216 in north England, and 1/100,000 in Germany.[10] Data suggest that by the age of 16 years, 71% of individuals will have developed some sign of HHT, rising to over 90% by the age of 40 years. In the past, a lower morbidity has been found in Asia; however, an investigation in 2002 performed by Dakeishi et al., found the morbidity in a county in north Japan, was 1/5,000-8,000, which was similar to that in West.[11] It was reported that about 20% had no family history, two cases in our study denied their family history. The common presentation of this syndrome is recurrent epistaxis caused by nasal mucocutaneous telangiectasia (90%), and others may present with telangiectasia of face, lips, limbs, and trunk.[12] In our study, pigmentation and telangiectasia were found in the skin over the upper abdomen in one case (Case#2). HHT patients may develop hemorrhage in gastric and intestinal tract in 20%, central nervous system arteriovenous malformation (AVM) in 20%, resulting in stroke and cranial abscess. Presence of the variation of the symptoms of HHT, in members of a single family, indicates that the personal phenotype depends on other factors, besides the inheritance. The main feature of the disease are multi-telangiectasia in the skin, mucosa, and internal organs with a trend of hemorrhage. Its clinical manifestations depend on the organ in which abnormal vessels are present.[13]
The basic pathology of Osler-Rendu-Weber syndrome involves the vascular wall of arteriole, veinlet, and capillaries. The walls become brittle and thin, lack elastofibrosis and smooth muscular layer, resulting in the dilation of the vascular lumen and the AVMs. This defect in the wall of the vessels predisposes these arties to rupture. There are three vascular dysplasia patterns of HHT: AVM (lung, brain), telangiectasia (mucosa, dermis, and visceral involvement), and pseudoaneurysms. The telangiectasias of varying extent were seen in our three cases.
It was estimated that at least 30% of HHT patients have hepatic lesions, and the morbidity from liver involvement is about 8-10%.[14] Liver manifestations in HHT include cirrhosis, hepatic fibrosis, and subsequent encephalopathy, esophagus varices and congestive heart failure. Presence of large AVMs results in encephalopathy and cardiac failure.
The measurement of hepatic volume can be used not only to quantitatively evaluate the configuration of the liver, but also to reflect the liver function indirectly, which has a potential to be regarded as a good clinical method to assess prognosis and the preservation of hepatic function, to choose suitable surgery, especially for the transplantation. The volume of the liver can reflect the changes of the dimension of the cells in parenchyma, and the blood perfusion and the metabolic energy. A study proved that the dimension of the liver with CT was similar to that of the real liver.[15] Hiroshige et al.,[16] found that spiral multislice CT was better than normal CT due to higher accuracy for the measuring the volume of liver with the three-dimensional construction. We also found the abnormal volume of the liver in three cases. In Case 1 and 2, we thought that the increasing volume was associated with arteriovenous shunts, telangiectasia, and hepatic congestion. In Case 3, the total volume was not enlarged; however, the caudate lobe was enlarged.
Surgery has limited role due to recurrent disease, but may be useful for emergency control of hemorrhage. Case 1 was operated because of portal hypertension which had a potential of bleeding. Surgery confirmed the CT findings of aneurysm-like dilated vessels, hepatomegaly, and cirrhosis. The left, right hepatic arteries and the vessels in porta hepatis were ligated, and a year of follow-up with clinical examination and imaging detection found that there was no recurrence. Case 2 was not suitable for the surgery. Liver transplantation and transcatheter embolotherapy are alternate treatments for patients with HHT.
In conclusion, CT is an accurate method for diagnosis and follow-up for Osler-Rendu-Weber syndrome. It can be used to follow-up patients after surgery for liver telangiectasias.
ACKNOWLEDGMENTS
This study was supported by the National Natural Science Foundation of China (No. 30970991), and the Natural Science Foundation of Shandong Province (No. Y2007C043).
Source of Support: Nil
Conflict of Interest: None declared.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2012/2/1/26/96541
REFERENCES
- Hereditary Haemorrhagic Telangiectasia (Rendu-Osler-Weber Disease) Lancet. 2003;362:1490-4.
- [Google Scholar]
- Screening family members of patients with hereditary haemorrhagic telangiectasia. Am J Med. 1995;99:519-24.
- [Google Scholar]
- MR of hereditary hemorrhagic telangiectasia: Prevalence and spectrum of cerebrovascular malformations. Am J Neuroradiol. 1998;19:477-84.
- [Google Scholar]
- Hepatic involvement in hereditary hemorrhagic telangiectasia: Clinical, radiological and hemodynamic studies of 11 cases. Gastroenterology. 1993;105:482-7.
- [Google Scholar]
- Liver involvement in hereditary hemorrhagic telangiectasia. Acta Gastroenterol Latinoam. 2011;41:225-9.
- [Google Scholar]
- Liver disease in patients with hereditary hemorrhagic telangiectasia. N Engl J Med. 2000;342:931-6.
- [Google Scholar]
- Improved diagnosis of hepatic perfusion disorders: Value of hepatic arterial phase imaging during helical CT. RadioGraphics. 2001;21:65-81.
- [Google Scholar]
- Accurate preoperative estimation of liver-graft volumetry using three-dimensional computed tomography. Transplantation. 2003;75:1561-4.
- [Google Scholar]
- Rendu-0sler-Weber disease: Experience with 56 patients. Ann Ital Med Int. 2002;17:173-9.
- [Google Scholar]
- Genetic epidemiology of hereditary hemorrhagic telangiectasia in a local comm unity in the northern part of Japan. Hum Mutat. 2002;19:140-8.
- [Google Scholar]
- Genetic heterogeneity in hereditary haemorrhages telangiectasia: Possible correlation with clinical phenotype. J Med Genet. 1994;3l:927-32.
- [Google Scholar]
- Linkage of hereditary haemorrhagic telangiectasia to chromosome 9q34 and evidence for locus heterogeneity. J Med Genet. 1994;31:933-6.
- [Google Scholar]
- Hereditary haemorrhagic telangiectasia with extensive liver involvement is not caused by either HHT1 or HHT2. J Med Genet. 1996;33:441-3.
- [Google Scholar]
- Graft size assessment and analysis of donors for living donor liver transplantation using right lobe. Transplantation. 2002;71:1407-13.
- [Google Scholar]
- Accurate preoperative estimation of liver-graft volumetry using three-dimensional computed tomography. Transplantation. 2003;75:1561-4.
- [Google Scholar]






