Translate this page into:
Arterial Tortuosity Syndrome: An Approach through Imaging Perspective
Address for correspondence: Dr. Venkatraman Bhat, 309, Greenwoods Apartment, Royal Gardenia, Bommasandra, Bangalore - 560 099, Karnataka, India. E-mail: bvenkatraman@gmail.com
-
Received: ,
Accepted: ,
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
This pictorial illustration demonstrates various aspects of arterial tortuosity syndrome (ATS) obtained predominantly from a multiple detector computed tomography (MDCT) examination of a patient. In addition, a comprehensive review of typical multi-modality imaging observations in patients with ATS is presented along with a description of a few imaging signs. Non-invasively obtained, conclusive information is required in patients with ATS in view of the fragile vascular structures involved. An amazing wealth of information can be obtained by reviewing the volumetric data sets of MDCT examination. In the context of incomplete clinical information or remote reading of radiographic examination with inadequate clinical details, ability to “image data mine” the hidden, unexplored information may be vastly useful. The role of MDCT as a single modality of evaluation in ATS is highlighted.
Keywords
Aortic elongation sign
arterial tortuosity syndrome
cluster of vessels sign
image data mining
multiple detector computed tomography
meandering vessel sign
V sign
INTRODUCTION

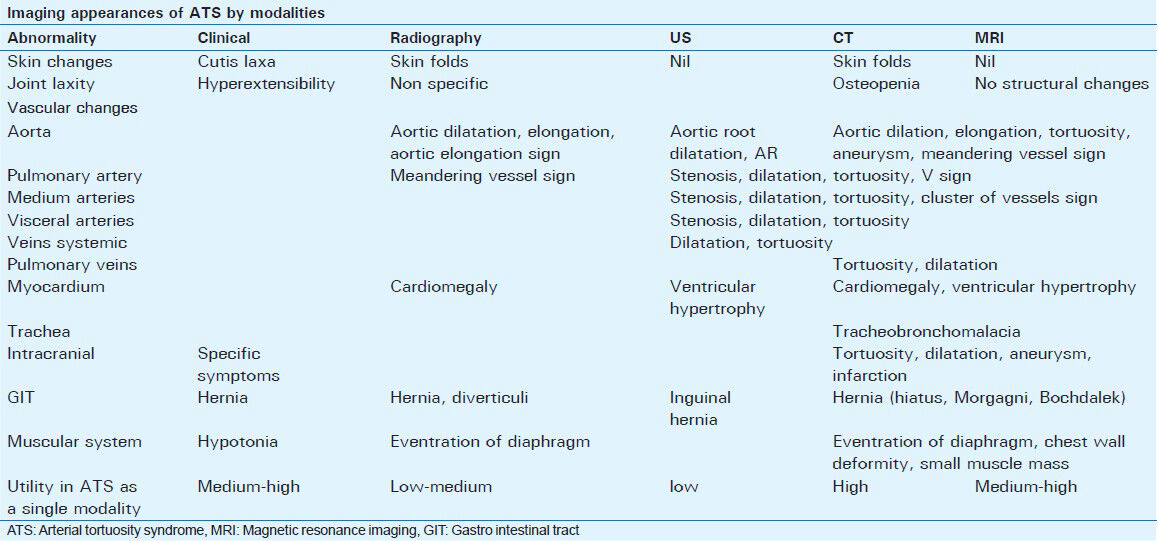
Arterial tortuosity syndrome (ATS) is a rare hereditary, autosomal recessive connective tissue disorder characterized by dysmorphic facial features, skin and joint laxity, tortuosity, elongation of the major arteries, vascular dilatation, stenosis, formation of aneurysm, pulmonary artery stenosis, and bowel hernia and rupture. Large and medium-sized arteries are mostly affected. Cardiomegaly with ventricular hypertrophy is seen in most patients. Bony changes like scoliosis, arachnodactyly, and chest wall deformity are also well-known associations.[12]
Many entities with inherited defects of connective tissue like Marfan syndrome (fibrillin-1), Williams Beuren syndrome (elastin), Ehlers Danlos syndrome (Type III collagen), and hereditary cutis laxa syndromes (elastin and fibulin-4 and -5) are known to present with very similar vascular and connective tissue changes. Relatively rarer conditions like ATS, Loeys–Dietz Syndrome (LDS), and autosomal recessive cutis laxa (ARCL) Type I and lethal syndromic vasculopathy associated with a novel mutation in FBLN4 are associated with more extensive vascular changes.[3]
ATS is an autosomal recessive disorder attributed to mutations in the SLC2A10 gene (chromosome 20q13).[45] LDS is an autosomal dominant disorder associated with heterozygous mutations in genes encoding transforming growth factor beta receptors (TGFBR) 1 and 2.[6] Perturbations in the TGF beta pathways are believed to contribute to the pathogenesis and severity of these disorders.[3]
The syndromes ATS and LDS are reported to have a natural history allowing survival of several patients to adulthood.[6] ARCL Type I (mutations of fibulin-4 and fibulin-4) is characterized by cutis laxa, pulmonary emphysema, umbilical and inguinal hernias, and gastrointestinal (GI) and vesico-urinary tract diverticuli. All these disorders have characteristic phenotypic features with a common feature of characteristic dilatation, elongation, and tortuosity of the large and medium-sized arteries.[36] Prior to the availability of genetic and molecular analysis, many of these patients were detected incidentally during radiographic examination. The purpose of this study is to illustrate cases from the imaging perspective, clustering subtle observations to build an image-based proof of ATS. Only a few studies in recent years have approached ATS from an imaging perspective. Advances in genetic and molecular diagnostics are redefining medical practice by identifying disease at a pre-clinical stage or at a stage when clinical and imaging appearances had not completely evolved, thus altering the conventional role of diagnostic imaging.
Due to intrinsic defect in the vascular collagen, blood vessel tends to show increased fragility and tendency for rupture. Hence, in the evaluation of these patients, invasive diagnostic procedures have been replaced by more non-invasive modalities like sonography, magnetic resonance imaging (MRI), and multidetector computed tomography (MDCT). Patients present clinically in different contexts varying from asymptomatic presentation to specific system-related symptoms, for example, a patient may present with cardiac failure and cardiovascular involvement or with gastroesophageal reflux/obstruction with hiatus hernia. Occasionally close relatives of patients with established ATS are referred for detailed investigation to demonstrate subtle imaging signs.
Sonographic evaluation can lead to a diagnosis of ATS when vascular tortuosity is seen in accessible regions. However, it fails to provide a global view of the vessels. Hence, as a comprehensive study, sonography fails to achieve its objective in the evaluation of ATS. However, it retains its valuable role in cardiac assessment and, at times, in evaluation of the large blood vessels. MRI has many advantages in demonstrating vascular changes, soft tissue changes secondary to internal and external hernia, and more specific vascular and structural intracranial complications of the ATS. Duration of the study and need for prolonged sedation limit the universal use of MRI techniques. Signs of airway compression, relation of vascular structure to lung and bowel, demonstration of lesions like hiatus hernia and inguinal hernia are not best achieved by MRI. MDCT evaluation addresses some of the limitations of ultrasound and MRI at the cost of radiation- and contrast-related complications. Hence, careful attention to technique in order to reduce radiation dose is necessary in these patients who may need repeated follow-up examinations. Emergence of new CT units with lower radiation dose, sensitive detector, and image processing technology are likely to overcome some of these disadvantages. However, on the whole, MDCT evaluation appears to be the best choice at the moment for rapid and comprehensive evaluation of ATS.[7] Illustrations of this work are based on sporadic cases with classical clinical signs and imaging observations and from the cohort of patients with mutations in the SLC2A10 gene (chromosome 20q13).[25] Most of the MDCT illustrations in this essay belong to a single patient, emphasizing the wealth of information obtained from a single MDCT study. Imaging signs described in the illustration vary from early signs to fairly classic signs of advanced disease. Understanding of these early signs may help radiologists detect patients with this disease who are clinically asymptomatic. Observations and conclusion of this presentation are based on the evaluation of 17 patients. Presentation of most patients was non-specific, and included failure to gain weight, fatigability, feeding difficulty, and vomiting. On clinical evaluation, facial dysmorphic features (micrognathia, elongated face, and epicanthal folds) and cardiac murmurs were noted, leading to echocardiography or chest radiography. Patients showed joint hypermobility and skin hyperextensibility. Parents were inter-related, and there was history of consanguinity in one cohort of patients. A single patient was detected on a prenatal scan with cardiac enlargement, leading to postnatal follow-up and diagnosis. One patient succumbed to cardiac failure, and the rest are on follow-up. Patients with hiatus hernia with esophageal symptoms were managed with ante-reflux medication. Surgical correction of hiatus hernia was performed successfully in one patient.
Examples shown follow a system-based illustrations depicting various component of ATS. Various manifestations of ATS in relation to different systems and the usefulness of specific imaging modality are presented in Table 1.
IMAGING OBSERVATION IN ATS
Skin laxity
Skin laxity is a clinical sign that suggests the possibility of a connective tissue disease. Though usually a clinical observation, occasionally direct and indirect signs are visualized on radiograph [Figure 1]. Plain radiographic examination of the chest demonstrates linear shadows overlying the lung fields. These shadows are due to shadows of exaggerated skin infolding. Sometimes this observation of a shadow can mislead the radiologist to erroneously identifying it as a case of pneumothorax. Surface-rendered evaluation of MDCT data can clearly visualize the skin folding. Such striking images are illustrated in Figure 2.

- Case 1: 7-month-old female child with poor weight gain and suspected cardiac disease was diagnosed with ATS. Frontal chest radiograph demonstrates multiple linear shadows overlying the right lung field due to excessive skin folding (black arrows). Also, there is retrocardiac lucency due to diaphragmatic hernia (empty arrow).

- Case 1: 7-month-old female with poor weight gain and suspected cardiac disease was diagnosed with ATS. Surface-rendered 3-D reconstruction (a and b) of the same patient illustrates excessive skin laxity leading to multiple redundant skin folds. Changes are striking in the back, axillary regions, and lower abdomen.
Skeletal deformity
Bony changes observed in ATS include arachnodactyly, chest wall deformity, joint hyperextensibility, and soft tissue contractures. Thoracic deformities are common in patients with ATS. Frequently observed deformities include kyphoscoliosis, pectus carinatum, and pectus excavatum [Figures 3 and 4]. Evaluation of MDCT data set for bone anomaly is greatly facilitated by various options in the 3-D processing software. Also, bony configurations like modeling abnormalities and bone thinning are illustrated.

- Case 1: 7-month-old female with poor weight gain and suspected cardiac disease was diagnosed with ATS. Surface-rendered 3-D image in (a) frontal projection demonstrates depression in chest wall, pectus excavatum deformity (black arrow). (b) Coronal contrast-enhanced sagittal CT image shows a depression below the sternum (white arrow).

- Case 1: 7-month-old female with poor weight gain and suspected cardiac disease was diagnosed with ATS. Frontal 3-D reconstructed view of spine reveals mild dorsal scoliosis with right-sided convexity. No malformation of the vertebral bodies was visualized. Ribs demonstrate a normal configuration.
Vascular tortuosity
Under the category of large vessel involvement, most striking observations are noted in the tortuosity and dilatation of the aortic arch. Findings range from subtle elongation of the aortic arch (aortic elongation sign) [Table 2 and Figure 5] to gross meandering of the aorta [Table 2 and Figures 6–8], almost reaching the lateral thoracic wall (meandering vessels sign)[8] [Figure 9]. The proximal arch up to the level of ductus shows marked tortuosity toward the right side, whereas aorta distal to the ductal position shows a tendency to extend to the left hemithorax. Areas of relative narrowing or kinking are observed at the level of the ductus and at the extreme bends of the curvature. The extent of vascular changes in different locations of vascular structures is attributed to the difference in distribution and amount of elastin in ATS patients. Secondary displacement of the airway and esophagus was noted in the patient presented in Figure 10. Airway compression is not very common; occasionally it may be seen leading to bronchial compression and lung collapse [Figure 8]. Tracheobronchomalacia is occasionally observed. Vascular changes could lead to aortic occlusion, aneurysm, and rupture into the pleural space, leading to a fatal outcome. On review of the existing literature, vascular calcification appears to be distinctively uncommon in ATS patients.


- Radiological signs in arterial tortuosity syndrome: Aortic elongation sign. (a and b) 4-month-old female with dysmorphic features and abnormal echo was diagnosed with ATS. Elongated tortuous aorta projects laterally (arrow). Sign is particularly useful in younger patient where the aortic knuckle is normally inconspicuous.

- Case 1: 7-month-old female with poor weight gain and suspected cardiac disease was diagnosed with ATS. (a and b) Maximal intensity projection images in coronal orientation show gross elongation, tortuosity of the thoracic aorta (empty arrow). Pulmonary artery is elongated (black arrow). (b) Tortuosity and dilatation are also noted in the proximal part of great vessels (empty arrow).
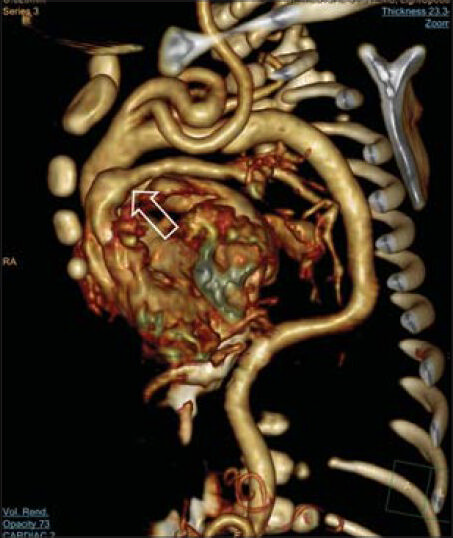
- Case 1: 7-month-old female with poor weight gain and suspected cardiac disease was diagnosed with ATS. Global view of 3-D rendered image of aorta in thoracic and abdominal part with extremely tortuous course of the thoracic aorta. Uneven caliber of the pulmonary artery is observed (empty arrow).

- Case 2: 1-month-old female with prenatal suspicion of cardiac disease was postnatally diagnosed with ATS. a) Coronal contrast-enhanced CT shows gross dilatation and tortuosity of thoracic and abdominal aorta (arrows). Incidentally there is left lung collapse due to bronchial compression. (b) 3-D volume-rendered image shows the global sign of gross aortic dilatation and tortuosity (arrows). At the areas of kinks, there is significant luminal narrowing (triangles).

- Radiological signs in arterial tortuosity syndrome: Meandering vessels sign. (a) Case 1. 7-month-female with poor weight gain and suspected cardiac disease was diagnosed with ATS. Coronal CT image shows lateral location of the tortuous aorta (empty arrows) reaching to mid-thoracic cavity. (b) 1-month-old female with prenatal suspicion of cardiac disease was postnatally diagnosed with ATS. CT image illustrates location of the meandering aorta up to the chest wall (empty arrows).

- Case 1: 7-month-old female presented with poor weight gain and suspected cardiac disease was diagnosed with ATS. (a) Coronal reconstruction of contrast-enhanced CT image shows splaying of the subcarinal angle (black arrows) due to cardiomegaly and a Bochdalek's hernia (empty arrow). (b) 3-D rendered image of airways does not show significant compression or indentation by the tortuous vessels.
The other group of vessels affected in ATS is that of pulmonary arteries which tend to show areas of narrowing and dilatation. Relative narrowing at the origin and early division of the main pulmonary artery lead to “V” (on coronal images) or “inverted V” (on axial images) configuration of the pulmonary bifurcation [Table 2 and Figures 11, 12]. This appearance is observed on echocardiography (personal communication) as well as MDCT and MRI. MRI plays a useful role in imaging of thoracic vascular structures, in co-operative patients, without exposure to ionizing radiation [Figures 13 and 14]. Significant stenosis with hemodynamic consequence is seen in more severe lesions. Peripheral pulmonary vessels, arteries, and veins, also show variation in caliber.

- Case 1: 7-month-old female with poor weight gain and suspected cardiac disease was diagnosed with ATS. 3-D rendered view of the pulmonary arteries seen from the posterior aspect demonstrates an early branching of the pulmonary artery (empty arrow) with uneven caliber of right and left pulmonary arteries.

- Radiological signs in arterial tortuosity syndrome: “V” sign of pulmonary artery. (a) Case 1: 7-month-old female with poor weight gain and suspected cardiac disease was diagnosed with ATS. Modified axial contrast-enhanced CT image demonstrates inverted V shape configuration of the pulmonary artery (black arrow) bifurcation and relative narrowing of proximal parts of right and left pulmonary arteries (arrows). (b) 4-month-old female with dysmorphic features and abnormal echo was diagnosed with ATS. T1-weighted MRI examination demonstrates the same configuration and narrow pulmonary arteries (arrows).

- Case 3: 4-month-old female with dysmorphic features and abnormal echo was diagnosed with ATS. Frontal chest radiograph classically demonstrates elongation of the aorta (white arrow), left diaphragmatic hernia (empty arrow), and dilatation and tortuosity of the right pulmonary artery (white triangle).

- Case 3: 4-month-old female with dysmorphic features and abnormal echo was diagnosed with ATS (same patient as in Figure 13). T1W MR images (a) in the sagittal orientation show the dilated pulmonary artery (white arrow) tortuous great vessels (black triangle) and grossly tortuous aorta. (b) Axial images show dilated, laterally displaced aortic arch (empty arrow). (c) Additional view at the level of pulmonary artery reveals dilated main pulmonary artery (white arrow) showing early bifurcation and rapid tapering of the caliber.
Involvement of pulmonary veins is not commonly described in ATS. However, tortuosity and uneven caliber can be occasionally seen involving the pulmonary veins [Figure 15].

- Case 1: 7-month-old female with poor weight gain and suspected cardiac disease was diagnosed with ATS. Coronal reconstruction of contrastenhanced CT image illustrates the right and left inferior pulmonary veins (empty arrows). Tortuosity and uneven caliber is also noted in both veins.
Peripheral arteries
Changes in the caliber and significant areas of widening and narrowing are seen in medium-sized and/or peripheral arteries. These changes are evident in the origin of great vessels and extend to peripheral vessels giving rise to a beaded appearance [Figures 16–18]. Gross tortuosity leading to altered course, and multiple folding and crowding of the arteries produce a classic sign of multiple clusters of vessels in the coronal and axial contrast-enhanced MDCT study of the thorax (cluster of vessels sign) [Table 2 and Figure 19]. Upper limb examination is generally included partially in the thoracic study, whereas lower limb arteries are not adequately evaluated. Involvement of neck and intracranial arteries is often observed, and is best evaluated by time-of-flight (TOF) and contrast-enhanced MR angiography [Figures 20 and 21]. Intracranial aneurysms are known in patients with ATS.[9] Visceral and abdominal arteries are also part of the vascular structures affected in the disease [Figure 22]. Kinking of the arteries is seen, particularly in celiac and superior mesenteric arteries [Figure 23]. Imaging attempts for vascular studies using femoral access show high complication rates. Venous access for CT examination also can be difficult. Complications include extravasations of contrast, vessel rupture, vessel destruction, pseudo-aneurysm formation, and vascular occlusion.

- Case 1: 7-month-old female with poor weight gain and suspected cardiac disease was diagnosed with ATS. 3-D rendered image of the vascular structures of the same patient shows extreme tortuosity and meandering course of the innominate (arrow) and left common carotid arteries (empty arrow). Incidentally, tortuosity of the segments of the subclavian arteries is also illustrated.

- Case 1: 7-month-old female with poor weight gain and suspected cardiac disease was diagnosed with ATS. 3-D rendered view of the left subclavian artery shows slightly tortuous course with uneven caliber (empty arrow). Tortuosity is particularly visible in the axillary and mid-arm region.

- Case 1: 7-month-old female with poor weight gain and suspected cardiac disease was diagnosed with ATS. 3-D rendered view of the right subclavian shows similar changes with a gross kink in the axillary region (empty arrow). Vascular changes are also seen in the small arteries like internal mammary (black arrows) which show beaded appearance.

- Radiological signs in arterial tortuosity syndrome: Cluster of vessels sign. (a) Case 1: 7-month-old female with poor weight gain and suspected cardiac disease was diagnosed with ATS. Sagittal CT reconstruction of contrast-enhanced examination reveals multiple rounded vascular structures in the superior mediastinum (arrows). (b) 4-month-old female with dysmorphic features and abnormal echo diagnosed with ATS. T1-weighted sagittal MR image shows multiple areas of flow voids corresponding to multiple cross-sections of tortuous vessels.

- 4-month-old male with abnormal facial features and cardiomegaly was diagnosed with ATS. Frontal chest radiograph demonstrates ectatic, laterally displaced aortic knuckle (white arrow). There is left ventricular cardiomegaly. Lungs are unremarkable.

- 9-year-old male with dysmorphic features and abnormal pulmonary arteries on echo was diagnosed with ATS. Time-of-flight MR images (a) in axial orientation show the gross tortuosity of the internal carotids in the paracavernous regions (white arrow). (b) Axial projection of the intracranial vessels with a TOF image demonstrates vascular tortuosity involving the carotids, basilar artery (empty arrow), and middle cerebral arteries (triangles).

- Case 1: 7-month-old female with poor weight gain and suspected cardiac disease was diagnosed with ATS. 3-D rendered image of the abdominal aorta and visceral arteries shows tortuosity in the superior mesenteric artery (empty arrow). Similar changes are also noted in the proximal part of the left renal artery (white arrow). Tortuosity without significant dilatation is seen in the iliac arteries (white triangles).

- Case 1: 7-month-old female with poor weight gain and suspected cardiac disease was diagnosed with ATS (same patient as in Figure 18). 3-D view of the abdominal aorta in the lateral projection demonstrates a kink in the proximal superior mesenteric artery (white arrow). More distally uneven caliber of superior mesenteric artery is shown with areas of multiple focal narrowing (empty arrows).
Gastroesophageal system
Esophageal dilatation and elongation is one of the frequent associations. Hiatus hernia [Figure 24], Morgagni hernia, and rarely a Bochdalek's hernia [Figure 25] are part of the disease spectrum. GI contrast studies may demonstrate diverticulum in duodenum and jejunum. Reflux in association with hiatus hernia is seen in vast majority of these patients.

- 4-month-old male with feeding difficulty and failure to gain weight was diagnosed with ATS. Barium studies (a and b) demonstrate a large, centrally located mixed type of hiatus hernia (empty arrows).

- Case 1: 7-month-old female with poor weight gain and suspected cardiac disease was diagnosed with ATS. (a) Coronal contrast-enhanced CT image reveals grossly displaced esophagus (empty arrow) and gastroesophageal junction. There is a large adjacent Bochdalek's hernia (black arrow). (b) Axial CT image shows lateral displacement of esophagus (arrow). (c) Sagittal CT reconstruction shows posteriorly located Bochdalek's hernia (black arrow).
Diaphragmatic eventration, diaphragmatic humps [Figure 26], hernia, and inguinal hernia occur due to hypotonia and defective connective tissue. Incidentally, CT examination may reveal decrease in the muscle mass and joint deformities.

- 4-month-old male child with abnormal facial features and failure to thrive was diagnosed with ATS. a) Frontal and b) left lateral chest radiograph reveals prominent, laterally displaced aorta (white arrow) and posteriorly located diaphragmatic eventrations (black arrows). Tortuous aorta is visualized as right retrocardiac density (triangle).
Most of the patients with mild, asymptomatic disease were on follow-up examination with yearly chest radiography, echocardiography, Doppler sonography, and MRI. MDCT was reserved for emergency presentation or when MRI was contraindicated. Ultrasound examination had a limited role in the follow-up evaluation of superficial vascular structures in the neck and extremities.
CONCLUSION
A spectrum of imaging features seen in a patient with ATS is illustrated in this report. A few imaging signs of ATS are described and highlighted with suitable illustrations. Awareness of early sign of arterial tortuosity and recognition of the described signs (aortic elongation sign and “V” sign of pulmonary artery) lead to early diagnosis of clinically asymptomatic cases of ATS. MDCT evaluation appears to be the best choice in the investigation of ATS patients. However, keeping the radiation dose to a minimum in the group of patients who may need multiple follow-up examinations is essential. Use of data mining of volumetric images from MDCT studies allows improved image interpretation and provides additional untapped facets of information, as exemplified in our case.
ACKNOWLEDGMENTS
The authors would like to acknowledge the contribution of Dr. Atiqa Abdul Wahab, Dr. Andrei Robida, Dr. Ibrahim Janahi, Dr. Abdulla Zeid, and Dr. Murugan Kuppusamy for providing insights into the problem and the constant clinical input required for this study. Special thanks to Dr. Paul C Salins for creative and administrative support. Also, the authors thank Philips Inc. for the workstation, Intellispace portal which was extensively used in the processing of volumetric CT data.
Available FREE in open access from: http://www.clinicalimagingscience.org/text.asp?2014/4/1/44/139734
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- Cardiovascular findings in a boy with arterial tortuosity syndrome: Case report and review of the literature. Turk J Pediatr. 2011;53:104-7.
- [Google Scholar]
- A new type of Ehlers-Danlos syndrome associated with tortuous systemic arteries in a large kindred from Qatar. Acta Paediatr. 2003;92:456-62.
- [Google Scholar]
- Characterization of a distinct lethal arteriopathy syndrome in twenty-two infants associated with an identical, novel mutation in FBLN4 gene, confirms fibulin-4 as a critical determinant of human vascular elastogenesis. Orphanet J Rare Dis. 2012;7:61.
- [Google Scholar]
- Mutations in the facilitative glucose transporter GLUT10 alter angiogenesis and cause arterial tortuosity syndrome. Nat Genet. 2006;38:452-7.
- [Google Scholar]
- Identification of a p. Ser81Arg encoding mutation in SLC2A10 gene of arterial tortuosity syndrome patients from 10 Qatari families. Clin Genet. 2008;74:189-93.
- [Google Scholar]
- A syndrome of altered cardiovascular, craniofacial, neurocognitive and skeletal development caused by mutations in TGFBR1 or TGFBR2. Nat Genet. 2005;37:275-81.
- [Google Scholar]
- Low-dose CT angiography for evaluation of great vessels and airway in arterial tortuosity syndrome. Eur Heart J Cardiovasc Imaging. 2012;13:1054.
- [Google Scholar]
- Meandering vessels: A sign of arterial tortuosity in plain chest radiography. Heartviews. 2008;9:24-6.
- [Google Scholar]
- Arterial tortuosity syndrome with multiple intracranial aneurysms: A case report. Arch Neurol. 2011;68:369-71.
- [Google Scholar]






